Chemistry is par excellence the science of transforming matter, and as such lies at the crossroads between the physical sciences and mathematics on the one hand, and the life sciences, earth sciences and materials sciences on the other. State-of-the-art laboratories, led by world-renowned researchers, are dedicated to a wide range of fields, from organic chemistry to materials chemistry, analytical chemistry and theoretical chemistry.
Find out more about the Chemistry Department
Spotlight
News

Three MSCA Doctoral Networks projects selected: a remarkable achievement for UNamur
Three MSCA Doctoral Networks projects selected: a remarkable achievement for UNamur
This is a great recognition of research at UNamur: three Marie Skłodowska-Curie Doctoral Networks (DN) projects have just been awarded, with a key contribution from researchers in Namur! The first, in chemistry, involves Professor Stéphane Vincent; the second, focused on ecosystem resilience, involves Professor Frédérik de Laender; and the third, in the field of photonics, benefits from the expertise of FNRS-qualified researcher Michaël Lobet.

For the MSCA Doctoral Networks 2025 call, 1,616 proposals were submitted and 141 were selected, representing a success rate of 9.6%. In this highly competitive environment, the selection of three projects involving UNamur sends a strong signal: it confirms the scientific excellence of Namur’s teams and their ability to build high-level international partnerships in support of doctoral training and innovation. Six doctoral dissertations will be eligible for funding.
Three projects, three cutting-edge topics
GlycoAxis – Understanding How the Gut Influences Brain Inflammation
Grant #101311186 from January 1, 2027, to December 31, 2031 – Project led by Stéphane Vincent – UNamur, Namur Research Institute for Life Sciences (NARILIS), in collaboration with 16 partners.
Coordination: Federico II University (Naples, Italy)

In many neurological diseases, both inflammation of the nervous system and imbalances in the gut microbiota are observed. GlycoAxis aims to go beyond simple correlations by identifying the molecular “messengers” that link the gut, the immune system, and the brain. The project focuses on complex sugars found on the surface of certain bacteria (glycans), which are suspected of playing a key role in immune activation and neuroinflammation. The goal: to better understand these mechanisms and pave the way for new diagnostic tools, imaging techniques, or biomarkers for brain health.
ReDiLeep – Strengthening ecosystem resilience through diverse responses
Grant # 101312530 from January 1, 2027, to December 31, 2031 – Project led by Frédérik de Laender – UNamur, Institute of Life, Earth and Environment (ILEE), in collaboration with 20 partners.
Coordination: Linköping University (Sweden).

In the face of climate change, pollution, and habitat fragmentation, some ecosystems weather the shocks… while others collapse. ReDiLeep focuses on a key driver of this resilience: response diversity—that is, the fact that different species (or ecological functions) do not all react in the same way to a disturbance. The project aims to better measure and model this mechanism in order to link research more directly to the needs of conservation, restoration, and public policy regarding biodiversity.
SPARK – programmable materials for controlling light at extremely high speeds
Grant # 101310184 from January 1, 2027, to December 31, 2031 – Project led by Michaël Lobet – UNamur, Namur Institute of Structured Matter (NISM), in collaboration with 7 partners.
Coordination: Eindhoven University of Technology (Netherlands)

Our digital communications rely on light: optical fibers, sensors, and photonic circuits capable of processing information. But with the explosion of data, the rise of AI, and the advent of ever-faster networks, it is becoming crucial to control light dynamically—much faster than is possible with current components, which are often “static.” SPARK is exploring a new approach: combining spatiotemporal metamaterials (nanoscale structures designed to shape light) with light that is itself “structured” in space and time. The result: reconfigurable photonic technologies for computing, imaging, and ultra-fast communications.
What are the Marie Skłodowska-Curie Doctoral Networks (MSCA-DN)?
In 1996, the European Union established the MSCA, a set of prestigious grants designed to fund research. The MSCA Doctoral Networks fund international networks that recruit and train doctoral students. Their goal is to combine high-level research with structured training, while promoting interdisciplinary and cross-sectoral collaboration as well as mobility within Europe and beyond.


Phoenix Capital sponsors research in synthetic organic chemistry
Phoenix Capital sponsors research in synthetic organic chemistry
At the Organic Chemistry Unit (UCO) of the Chemistry Department at UNamur, researchers study organic molecules with high added value. Lionel-Marie Van Geesbergen has just started a doctoral thesis under the supervision of Professor Steve Lanners. After only a month and a half of investigation, the PhD student already has a proof of concept. A very promising research project with numerous potential applications!

In chemistry, cycloaddition reactions are sometimes difficult to achieve. This is because two molecules mixed together often do not react with each other, as they encounter each other too rarely to produce an effective reaction. However, these reactions are fundamental in organic chemistry because they enable complex structures to be assembled quickly.
One way to get around this lack of reactivity is to physically bring the molecules closer together by connecting them with a bond that can be broken once it has done its job. This approach is called ‘tethering’. By applying this strategy, the two molecules are maintained in close proximity so that they have no choice but to react together.
A little organic chemistry
In the context of this project, the tethering strategy is applied to little-studied cycloaddition precursors: oxidopyridiniums. The interest of these compounds is that they allow rapid and selective access to nitrogen-containing polycyclic products, but their use without tethering is generally ineffective.
In general, nitrogen-containing polycyclic molecules are organic compounds in which nitrogen is present within the cyclic structure (heterocycles) or as a substituent. These molecules are ubiquitous in medicinal chemistry, biochemistry and agrochemistry. They play a key and fundamental structural role in biology, pharmacology and organic chemistry.
More specifically, in the context of this project, among the families of molecules that can be obtained in this way are, for example, tropanes, a family of bicyclic alkaloids obtained from natural sources, some of which (or their derivatives) are used as medicines. The well-known over-the-counter medication Buscopan belongs to this family.
If we want to go further, this is where the second objective of this project comes into play: applying the Beckmann rearrangement to the products obtained by cycloaddition. This allows a second nitrogen atom to be introduced into the structures and opens up prospects for the synthesis of phlegmadines, a group of natural products that have never been prepared by organic synthesis, even though their described biological properties are promising. More recently, in the field of oncology research, KRAS inhibitors containing dinitrogenated bicylic structures have been described.
But what's the point?
Taking a step back, the aim is to make chemical synthesis, and therefore the production of molecules in large quantities, more sustainable: target products are prepared more quickly and efficiently. This speeds up research while reducing the impact on the environment. The fewer steps involved in manufacturing a product, the less water, solvents, reagents and time are used: this means less energy consumption, less waste and lower costs, while also speeding up the process!
Lionel-Marie Van Geesbergen had already successfully investigated these reactions accelerated by stapling with oxygenated molecules during his master's thesis in chemistry at UNamur in the same laboratory. After only a month and a half of research, the doctoral student has already demonstrated the feasibility of his method with nitrogen molecules. Now that the approach has been validated, it can be developed to determine its scope and limitations.
A project supported by Phoenix Capital
This doctoral project will be supported for 4 years by the company Phoenix Capital. Headquartered in Italy (Verona) and active in Milan, Rome, Luxembourg, Lausanne, Houston and San Francisco, Phoenix Capital is a management consultancy, innovation, corporate finance hub also offering technological and operational services for business development and the streamlining of processes and projects.

As a driver of innovation and development in Europe and in the US, Phoenix Capital develops numerous synergies with universities in Italy and internationally, promoting excellence in education.
By supporting this research project, Phoenix Capital encourages scientific research and cutting-edge technologies developed by UNamur in the field of synthetic organic chemistry.
We are proud to support the four-year PhD program in Chemistry at the University of Namur. Investing in research is a long-term strategic choice for Phoenix Capital and is in line with our Ethical Manifesto in terms of supporting young talent. We are increasingly convinced of this, especially today given the international context in which we operate.

It is in universities that ideas are born that can improve people's lives, make supply chains more competitive, and accelerate the transition to sustainable production models. At the heart of this vision are young talents who cultivate a passion for science: researchers who, with curiosity and rigor, transform today's questions into tomorrow's solutions. Building bridges between universities and businesses means giving them tools, time, and trust. This is how we intend to contribute to a stronger, more inclusive, and more responsible innovation ecosystem.
Giovanna Saraconi - CEO Phoenix Group
The researchers at the Laboratory of Synthetic Organic Chemistry (COS)
Steve Lanners
Steve Lanners studied chemistry at Louis Pasteur University, then at ECPM in Strasbourg (class of 2001). After a research stay at ETH (with Prof. Erick Carreira) where he synthesised a fragment of the anti-cancer polyketide tedanolide, he completed his PhD at ECPM under the supervision of Prof. Guy Solladié and Dr. Gilles Hanquet (defended in 2005) to develop the synthesis of a polyketide with anti-mybacterial activity: pamamycin-607.

After three years of postdoctoral research in flow chemistry and natural product synthesis at the University of Cambridge with Prof. Steven Ley, he joined the University of Namur, where he took over the Organic Synthesis Chemistry (COS) laboratory to develop projects in natural product synthesis, new reaction development and medicinal chemistry, while holding various positions within the Chemistry Department and working to constantly improve the teaching of organic chemistry at UNamur.
Lionel-Marie Van Geesbergen
Lionel-Marie Van Geesbergen completed his bachelor's degree in June 2022 and decided to pursue a master's degree in chemical sciences at the University of Namur. During this master's programme, and more specifically during his thesis supervised by Prof. Steve Lanners, he participated in the development of a synthesis pathway for complex molecules using an innovative strategy: ‘temporary stapling’.

As part of his internship, he then collaborated with Syensqo on a project to valorise by-products from the polymer industry in the laboratory of Prof. Gwilherm Evano at the Free University of Brussels. These experiences enabled him to obtain his master's degree in June 2024. After graduating, he participated in the supervision and training of undergraduate students in pharmacy and biomedical sciences in chemistry, both during exercise sessions and practical work. In January 2026, he chose to return to Prof. Lanners' team to begin a doctoral thesis and continue the research he had started during his dissertation, focusing on the synthesis of complex nitrogenous molecules with high pharmaceutical potential.

Producing "green" hydrogen from water from the Meuse River? It's now possible!
Producing "green" hydrogen from water from the Meuse River? It's now possible!
At UNamur, research is not confined to laboratories. From physics to political science, robotics, biodiversity, law, AI, and health, researchers collaborate daily with numerous stakeholders in society. The goal? Transform ideas into concrete solutions to address current challenges.

Focus #2 | What if our rivers became a source of clean energy for the future?
An international team of chemistry researchers, led by Dr. Laroussi Chaabane and Prof. Bao-Lian Su, has just demonstrated that it is possible to produce "green" hydrogen using natural water and sunlight. These findings have been published in the prestigious Chemical Engineering Journal.
When sunlight becomes a source of clean energy
Faced with climate change, pollution, and energy shortages, the search for alternatives to fossil fuels has become a global priority in order to achieve carbon neutrality by 2050. Among the solutions being considered, green hydrogen appears to be a particularly promising energy carrier: it has a high energy density and can be produced without greenhouse gas emissions. Today, most of the world's hydrogen (around 87 million tons produced in 2020) is obtained through costly and polluting electrochemical processes, mainly used by the chemical industry or fuel cells. Hence the major interest in more sustainable methods.
Water photocatalysis: the "Holy Grail" of chemistry
Producing hydrogen and oxygen directly from water using light, a process known as photocatalysis of water, is often referred to as the "Holy Grail of chemistry" because it is so complex to master. At the University of Namur, researchers at the Laboratory of Inorganic Materials Chemistry (CMI), part of the Nanomaterials Chemistry Unit (UCNANO) and the Namur Institute of Structured Matter (NISM), have taken a decisive step forward. They have demonstrated that it is possible to use natural water, and no longer just ultrapure water, to produce green hydrogen under the action of sunlight.

The core of the process is based on an innovative photocatalyst, which acts as a kind of "chemical pair of scissors" capable of splitting water molecules into hydrogen and oxygen—an area in which the CMI laboratory has recognized expertise.
A 3D photocatalyst based on graphene and gold
The new material developed is a three-dimensional (3D) photocatalyst based on titanium oxide, graphene, and gold nanoparticles. This 3D architecture allows for better light absorption and more efficient generation of free electrons, which are essential for triggering the water dissociation reaction. One of the main challenges lies in the use of natural water, which contains minerals, salts, and organic compounds that can disrupt the process. To address this challenge, the researchers tested their device with water from several Belgian rivers: the Meuse, the Sambre, the Scheldt, and the Yser.
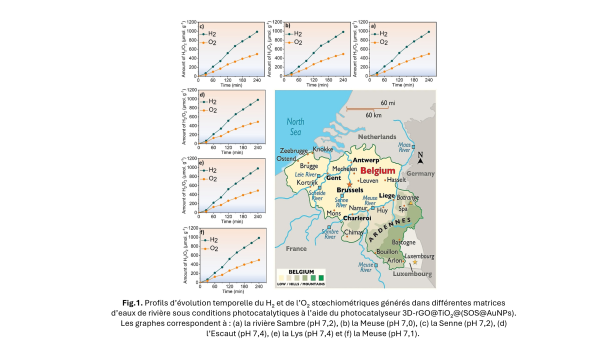
A remarkable result and a first in Belgium!
The performance achieved is almost equivalent to that measured with pure water.
This is a first in Belgium, opening up concrete prospects for the sustainable use of local natural resources!
The full article, "Synergistic four physical phenomena in a 3D photocatalyst for unprecedented overall water splitting," is available in open access.
International recognition
This scientific breakthrough also earned Dr. Laroussi Chaabane the award for best poster at the 4th International Colloids Conference (San Sebastián, Spain, July 2025), highlighting the impact and originality of this work.
An international research team
- University of Namur, Faculty of Sciences, UCNANO, Laboratory of Inorganic Materials Chemistry (CMI) and Namur Institute of Structured Matter (NISM), Belgium | Principal Investigator (PI) | Professor Bao Lian SU; Postdoctoral Researcher | Dr. Laroussi Chaabane
- Institute of Organic Chemistry, Phytochemistry Center, Academy of Sciences, Bulgaria
- Department of Organic Chemistry (MSc), Loyola Academy, India
- Free University of Brussels (ULB) and Flanders Make, Department of Applied Physics and Photonics, Brussels Photonics, Belgium
- University of Quebec in Montreal (UQAM), Department of Chemistry, Montreal, Quebec, Canada
- National Institute for Scientific Research - Energy Materials Telecommunications Center (INRS-EMT), Varennes, Quebec, Canada
- Wuhan University of Technology, National Laboratory for Advanced Technologies in Materials Synthesis and Processing, China
What next?
At this stage, the study constitutes proof of concept demonstrating the feasibility of the process. It illustrates the excellence of chemical engineering and nanomaterials research at UNamur, as well as its potential for sustainable energy applications. A new study is underway to evaluate the performance of the process with seawater, a key step towards large-scale green hydrogen production.
State-of-the-art equipment
The analyses carried out were made possible thanks to the equipment available at UNamur's Physico-Chemical Characterization (PC²), Electron Microscopy, and Material Synthesis, Irradiation, and Analysis (SIAM) technology platforms. UNamur's technology platforms house state-of-the-art equipment and are accessible to the scientific community as well as to industries and companies.
The authors would like to thank the Wallonia Public Service (SPW) for its ongoing commitment to scientific research and innovation in Wallonia, enabling UNamur to develop technological solutions with a significant societal and environmental impact.
From fundamental to applied research, UNamur demonstrates every day that research is a driver of transformation. Thanks to the commitment of its researchers, the support of its partners from all walks of life, funders, industrial partners, and a solid ecosystem of valorization, UNamur actively participates in shaping a society that is open to the world, more innovative, more responsible, and more sustainable.
To go further
This article complements our publication "Research and innovation: major assets for the industrial sector" taken from the Issues section of Omalius magazine #39 (December 2025).

Véronique Steukers, President of the Nickel Institute
Véronique Steukers, President of the Nickel Institute
A chemist by training, Véronique Steukers is now the first woman to head the global organization of nickel producers, the Nickel Institute. Her career path has taken her far from the laboratory and into the heart of an industry facing significant environmental, industrial, and social challenges. We meet her.

What makes nickel so essential today?
Nickel is a surprisingly versatile metal, valued for its strength and durability. This explains its presence in many everyday applications. It has been used for over 100 years in the manufacture of stainless steel, which can be found everywhere in our kitchens, particularly in sinks and cutlery. Nickel makes these utensils highly durable and more resistant to corrosion, particularly from household products. It is also found in certain infrastructures. The Atomium, for example, was completely covered with a layer of nickel-containing stainless steel after its original coating had aged. This guarantees several decades of resistance without degradation. Nickel is also essential for electric car batteries because it improves their energy density, as well as for many other renewable energy technologies. Finally, it is one of the most recycled metals and its importance is set to grow with the increasing return of end-of-life batteries.
What is the role of the Nickel Institute?
The Nickel Institute is a global organization based in Canada with offices on several continents. Its main mission is to promote responsible nickel sourcing and support the sustainable development of this industry. To this end, we have three complementary departments. The first, scientific, is composed of toxicologists specializing in human and environmental health, as well as a prevention advisor responsible for worker protection. The second focuses on public policy and sustainability by monitoring developments in international regulations. It develops methodologies, particularly for measuring carbon footprints. Finally, the third is dedicated to market development and ensures that the various applications of nickel are better known and that markets remain open globally.
You have just been appointed president of the Nickel Institute. What does this appointment mean for your career as a chemist?
It's a role I really wanted to achieve in my career. After studying at UNamur, spending a year studying industrial chemistry in Leuven, completing a PhD in England and a post-doctorate in the Netherlands, I no longer practiced chemistry in a laboratory, but my training has always accompanied my career. It gave me an analytical and critical mind, an understanding of chemical substances, their properties, and industrial processes. This has enabled me to communicate effectively with authorities and stakeholders throughout my career. I often explain that studying chemistry does not only lead to a career in the laboratory. It opens many doors and gives access to a multitude of career paths.
What environmental and societal challenges await the nickel industry?
The stakes are high, especially because this metal is essential to the energy transition. It is found in many climate-related technologies, such as electric car batteries, hydroelectricity, and wind turbines. But nickel remains a mining product, and its production is mainly located in developing countries where the environment, working conditions, and local communities are not always a priority. That is why the Nickel Institute works closely with authorities, companies, and other stakeholders to improve understanding of nickel and its risks. The goal is to ensure that those who produce or handle this metal are aware of best practices for managing the risks associated with extraction, production, and industrial use. The challenge, therefore, remains to guide the industry toward more responsible and sustainable practices.
What do you remember most about your time at UNamur?
I loved my years at UNamur. I am Flemish, and many of my friends did not understand why I chose Namur over Leuven, but I have never regretted it. The atmosphere was very friendly, and the professors and teaching assistants were very welcoming. So much so that we are still in touch, forty years after our first year studying chemistry!
Is there a particular memory you would like to share?
One of my fondest memories is the series of concerts we organized at the Arsenal for two years in a row. I played the piano and had the opportunity to perform a piece for four hands with Professor Jean-Marie André, as well as a trio with two other professors. It was something that came very naturally in Namur, thanks to the small size of the university, where everyone knew each other. I'm not sure I would have found that same closeness anywhere else.
What advice would you give to future chemists?
Don't hesitate! Studying chemistry opens many doors. Of course, you need to have a scientific mind, but it's a course of study that allows you to develop skills that are useful in many professions. I would also advise placing real importance on languages. In Belgium, mastering several languages is an essential asset for advancing in industry. I also notice that there are many more opportunities for women in scientific careers than there used to be. At the beginning of my professional life, I was often the only woman in the room, but today teams are much more mixed, even in heavy industries such as metals. The fact that I became the first female president of the Nickel Institute is quite encouraging.
This article is taken from the "Alumni" section of Omalius magazine #39 (December 2025).

Did you know?
February 11 is International Day of Women and Girls in Science. To mark the occasion, UNamur is organizing the sixth edition of its Women in Science conference. This annual event aims to promote women's and girls' access to science and technology and their full and equal participation. It highlights the important role of women in the scientific community and is an excellent opportunity to encourage and promote equal opportunities for all genders in science and technology.

Three MSCA Doctoral Networks projects selected: a remarkable achievement for UNamur
Three MSCA Doctoral Networks projects selected: a remarkable achievement for UNamur
This is a great recognition of research at UNamur: three Marie Skłodowska-Curie Doctoral Networks (DN) projects have just been awarded, with a key contribution from researchers in Namur! The first, in chemistry, involves Professor Stéphane Vincent; the second, focused on ecosystem resilience, involves Professor Frédérik de Laender; and the third, in the field of photonics, benefits from the expertise of FNRS-qualified researcher Michaël Lobet.

For the MSCA Doctoral Networks 2025 call, 1,616 proposals were submitted and 141 were selected, representing a success rate of 9.6%. In this highly competitive environment, the selection of three projects involving UNamur sends a strong signal: it confirms the scientific excellence of Namur’s teams and their ability to build high-level international partnerships in support of doctoral training and innovation. Six doctoral dissertations will be eligible for funding.
Three projects, three cutting-edge topics
GlycoAxis – Understanding How the Gut Influences Brain Inflammation
Grant #101311186 from January 1, 2027, to December 31, 2031 – Project led by Stéphane Vincent – UNamur, Namur Research Institute for Life Sciences (NARILIS), in collaboration with 16 partners.
Coordination: Federico II University (Naples, Italy)

In many neurological diseases, both inflammation of the nervous system and imbalances in the gut microbiota are observed. GlycoAxis aims to go beyond simple correlations by identifying the molecular “messengers” that link the gut, the immune system, and the brain. The project focuses on complex sugars found on the surface of certain bacteria (glycans), which are suspected of playing a key role in immune activation and neuroinflammation. The goal: to better understand these mechanisms and pave the way for new diagnostic tools, imaging techniques, or biomarkers for brain health.
ReDiLeep – Strengthening ecosystem resilience through diverse responses
Grant # 101312530 from January 1, 2027, to December 31, 2031 – Project led by Frédérik de Laender – UNamur, Institute of Life, Earth and Environment (ILEE), in collaboration with 20 partners.
Coordination: Linköping University (Sweden).

In the face of climate change, pollution, and habitat fragmentation, some ecosystems weather the shocks… while others collapse. ReDiLeep focuses on a key driver of this resilience: response diversity—that is, the fact that different species (or ecological functions) do not all react in the same way to a disturbance. The project aims to better measure and model this mechanism in order to link research more directly to the needs of conservation, restoration, and public policy regarding biodiversity.
SPARK – programmable materials for controlling light at extremely high speeds
Grant # 101310184 from January 1, 2027, to December 31, 2031 – Project led by Michaël Lobet – UNamur, Namur Institute of Structured Matter (NISM), in collaboration with 7 partners.
Coordination: Eindhoven University of Technology (Netherlands)

Our digital communications rely on light: optical fibers, sensors, and photonic circuits capable of processing information. But with the explosion of data, the rise of AI, and the advent of ever-faster networks, it is becoming crucial to control light dynamically—much faster than is possible with current components, which are often “static.” SPARK is exploring a new approach: combining spatiotemporal metamaterials (nanoscale structures designed to shape light) with light that is itself “structured” in space and time. The result: reconfigurable photonic technologies for computing, imaging, and ultra-fast communications.
What are the Marie Skłodowska-Curie Doctoral Networks (MSCA-DN)?
In 1996, the European Union established the MSCA, a set of prestigious grants designed to fund research. The MSCA Doctoral Networks fund international networks that recruit and train doctoral students. Their goal is to combine high-level research with structured training, while promoting interdisciplinary and cross-sectoral collaboration as well as mobility within Europe and beyond.


Phoenix Capital sponsors research in synthetic organic chemistry
Phoenix Capital sponsors research in synthetic organic chemistry
At the Organic Chemistry Unit (UCO) of the Chemistry Department at UNamur, researchers study organic molecules with high added value. Lionel-Marie Van Geesbergen has just started a doctoral thesis under the supervision of Professor Steve Lanners. After only a month and a half of investigation, the PhD student already has a proof of concept. A very promising research project with numerous potential applications!

In chemistry, cycloaddition reactions are sometimes difficult to achieve. This is because two molecules mixed together often do not react with each other, as they encounter each other too rarely to produce an effective reaction. However, these reactions are fundamental in organic chemistry because they enable complex structures to be assembled quickly.
One way to get around this lack of reactivity is to physically bring the molecules closer together by connecting them with a bond that can be broken once it has done its job. This approach is called ‘tethering’. By applying this strategy, the two molecules are maintained in close proximity so that they have no choice but to react together.
A little organic chemistry
In the context of this project, the tethering strategy is applied to little-studied cycloaddition precursors: oxidopyridiniums. The interest of these compounds is that they allow rapid and selective access to nitrogen-containing polycyclic products, but their use without tethering is generally ineffective.
In general, nitrogen-containing polycyclic molecules are organic compounds in which nitrogen is present within the cyclic structure (heterocycles) or as a substituent. These molecules are ubiquitous in medicinal chemistry, biochemistry and agrochemistry. They play a key and fundamental structural role in biology, pharmacology and organic chemistry.
More specifically, in the context of this project, among the families of molecules that can be obtained in this way are, for example, tropanes, a family of bicyclic alkaloids obtained from natural sources, some of which (or their derivatives) are used as medicines. The well-known over-the-counter medication Buscopan belongs to this family.
If we want to go further, this is where the second objective of this project comes into play: applying the Beckmann rearrangement to the products obtained by cycloaddition. This allows a second nitrogen atom to be introduced into the structures and opens up prospects for the synthesis of phlegmadines, a group of natural products that have never been prepared by organic synthesis, even though their described biological properties are promising. More recently, in the field of oncology research, KRAS inhibitors containing dinitrogenated bicylic structures have been described.
But what's the point?
Taking a step back, the aim is to make chemical synthesis, and therefore the production of molecules in large quantities, more sustainable: target products are prepared more quickly and efficiently. This speeds up research while reducing the impact on the environment. The fewer steps involved in manufacturing a product, the less water, solvents, reagents and time are used: this means less energy consumption, less waste and lower costs, while also speeding up the process!
Lionel-Marie Van Geesbergen had already successfully investigated these reactions accelerated by stapling with oxygenated molecules during his master's thesis in chemistry at UNamur in the same laboratory. After only a month and a half of research, the doctoral student has already demonstrated the feasibility of his method with nitrogen molecules. Now that the approach has been validated, it can be developed to determine its scope and limitations.
A project supported by Phoenix Capital
This doctoral project will be supported for 4 years by the company Phoenix Capital. Headquartered in Italy (Verona) and active in Milan, Rome, Luxembourg, Lausanne, Houston and San Francisco, Phoenix Capital is a management consultancy, innovation, corporate finance hub also offering technological and operational services for business development and the streamlining of processes and projects.

As a driver of innovation and development in Europe and in the US, Phoenix Capital develops numerous synergies with universities in Italy and internationally, promoting excellence in education.
By supporting this research project, Phoenix Capital encourages scientific research and cutting-edge technologies developed by UNamur in the field of synthetic organic chemistry.
We are proud to support the four-year PhD program in Chemistry at the University of Namur. Investing in research is a long-term strategic choice for Phoenix Capital and is in line with our Ethical Manifesto in terms of supporting young talent. We are increasingly convinced of this, especially today given the international context in which we operate.

It is in universities that ideas are born that can improve people's lives, make supply chains more competitive, and accelerate the transition to sustainable production models. At the heart of this vision are young talents who cultivate a passion for science: researchers who, with curiosity and rigor, transform today's questions into tomorrow's solutions. Building bridges between universities and businesses means giving them tools, time, and trust. This is how we intend to contribute to a stronger, more inclusive, and more responsible innovation ecosystem.
Giovanna Saraconi - CEO Phoenix Group
The researchers at the Laboratory of Synthetic Organic Chemistry (COS)
Steve Lanners
Steve Lanners studied chemistry at Louis Pasteur University, then at ECPM in Strasbourg (class of 2001). After a research stay at ETH (with Prof. Erick Carreira) where he synthesised a fragment of the anti-cancer polyketide tedanolide, he completed his PhD at ECPM under the supervision of Prof. Guy Solladié and Dr. Gilles Hanquet (defended in 2005) to develop the synthesis of a polyketide with anti-mybacterial activity: pamamycin-607.

After three years of postdoctoral research in flow chemistry and natural product synthesis at the University of Cambridge with Prof. Steven Ley, he joined the University of Namur, where he took over the Organic Synthesis Chemistry (COS) laboratory to develop projects in natural product synthesis, new reaction development and medicinal chemistry, while holding various positions within the Chemistry Department and working to constantly improve the teaching of organic chemistry at UNamur.
Lionel-Marie Van Geesbergen
Lionel-Marie Van Geesbergen completed his bachelor's degree in June 2022 and decided to pursue a master's degree in chemical sciences at the University of Namur. During this master's programme, and more specifically during his thesis supervised by Prof. Steve Lanners, he participated in the development of a synthesis pathway for complex molecules using an innovative strategy: ‘temporary stapling’.

As part of his internship, he then collaborated with Syensqo on a project to valorise by-products from the polymer industry in the laboratory of Prof. Gwilherm Evano at the Free University of Brussels. These experiences enabled him to obtain his master's degree in June 2024. After graduating, he participated in the supervision and training of undergraduate students in pharmacy and biomedical sciences in chemistry, both during exercise sessions and practical work. In January 2026, he chose to return to Prof. Lanners' team to begin a doctoral thesis and continue the research he had started during his dissertation, focusing on the synthesis of complex nitrogenous molecules with high pharmaceutical potential.

Producing "green" hydrogen from water from the Meuse River? It's now possible!
Producing "green" hydrogen from water from the Meuse River? It's now possible!
At UNamur, research is not confined to laboratories. From physics to political science, robotics, biodiversity, law, AI, and health, researchers collaborate daily with numerous stakeholders in society. The goal? Transform ideas into concrete solutions to address current challenges.

Focus #2 | What if our rivers became a source of clean energy for the future?
An international team of chemistry researchers, led by Dr. Laroussi Chaabane and Prof. Bao-Lian Su, has just demonstrated that it is possible to produce "green" hydrogen using natural water and sunlight. These findings have been published in the prestigious Chemical Engineering Journal.
When sunlight becomes a source of clean energy
Faced with climate change, pollution, and energy shortages, the search for alternatives to fossil fuels has become a global priority in order to achieve carbon neutrality by 2050. Among the solutions being considered, green hydrogen appears to be a particularly promising energy carrier: it has a high energy density and can be produced without greenhouse gas emissions. Today, most of the world's hydrogen (around 87 million tons produced in 2020) is obtained through costly and polluting electrochemical processes, mainly used by the chemical industry or fuel cells. Hence the major interest in more sustainable methods.
Water photocatalysis: the "Holy Grail" of chemistry
Producing hydrogen and oxygen directly from water using light, a process known as photocatalysis of water, is often referred to as the "Holy Grail of chemistry" because it is so complex to master. At the University of Namur, researchers at the Laboratory of Inorganic Materials Chemistry (CMI), part of the Nanomaterials Chemistry Unit (UCNANO) and the Namur Institute of Structured Matter (NISM), have taken a decisive step forward. They have demonstrated that it is possible to use natural water, and no longer just ultrapure water, to produce green hydrogen under the action of sunlight.

The core of the process is based on an innovative photocatalyst, which acts as a kind of "chemical pair of scissors" capable of splitting water molecules into hydrogen and oxygen—an area in which the CMI laboratory has recognized expertise.
A 3D photocatalyst based on graphene and gold
The new material developed is a three-dimensional (3D) photocatalyst based on titanium oxide, graphene, and gold nanoparticles. This 3D architecture allows for better light absorption and more efficient generation of free electrons, which are essential for triggering the water dissociation reaction. One of the main challenges lies in the use of natural water, which contains minerals, salts, and organic compounds that can disrupt the process. To address this challenge, the researchers tested their device with water from several Belgian rivers: the Meuse, the Sambre, the Scheldt, and the Yser.

A remarkable result and a first in Belgium!
The performance achieved is almost equivalent to that measured with pure water.
This is a first in Belgium, opening up concrete prospects for the sustainable use of local natural resources!
The full article, "Synergistic four physical phenomena in a 3D photocatalyst for unprecedented overall water splitting," is available in open access.
International recognition
This scientific breakthrough also earned Dr. Laroussi Chaabane the award for best poster at the 4th International Colloids Conference (San Sebastián, Spain, July 2025), highlighting the impact and originality of this work.
An international research team
- University of Namur, Faculty of Sciences, UCNANO, Laboratory of Inorganic Materials Chemistry (CMI) and Namur Institute of Structured Matter (NISM), Belgium | Principal Investigator (PI) | Professor Bao Lian SU; Postdoctoral Researcher | Dr. Laroussi Chaabane
- Institute of Organic Chemistry, Phytochemistry Center, Academy of Sciences, Bulgaria
- Department of Organic Chemistry (MSc), Loyola Academy, India
- Free University of Brussels (ULB) and Flanders Make, Department of Applied Physics and Photonics, Brussels Photonics, Belgium
- University of Quebec in Montreal (UQAM), Department of Chemistry, Montreal, Quebec, Canada
- National Institute for Scientific Research - Energy Materials Telecommunications Center (INRS-EMT), Varennes, Quebec, Canada
- Wuhan University of Technology, National Laboratory for Advanced Technologies in Materials Synthesis and Processing, China
What next?
At this stage, the study constitutes proof of concept demonstrating the feasibility of the process. It illustrates the excellence of chemical engineering and nanomaterials research at UNamur, as well as its potential for sustainable energy applications. A new study is underway to evaluate the performance of the process with seawater, a key step towards large-scale green hydrogen production.
State-of-the-art equipment
The analyses carried out were made possible thanks to the equipment available at UNamur's Physico-Chemical Characterization (PC²), Electron Microscopy, and Material Synthesis, Irradiation, and Analysis (SIAM) technology platforms. UNamur's technology platforms house state-of-the-art equipment and are accessible to the scientific community as well as to industries and companies.
The authors would like to thank the Wallonia Public Service (SPW) for its ongoing commitment to scientific research and innovation in Wallonia, enabling UNamur to develop technological solutions with a significant societal and environmental impact.
From fundamental to applied research, UNamur demonstrates every day that research is a driver of transformation. Thanks to the commitment of its researchers, the support of its partners from all walks of life, funders, industrial partners, and a solid ecosystem of valorization, UNamur actively participates in shaping a society that is open to the world, more innovative, more responsible, and more sustainable.
To go further
This article complements our publication "Research and innovation: major assets for the industrial sector" taken from the Issues section of Omalius magazine #39 (December 2025).

Véronique Steukers, President of the Nickel Institute
Véronique Steukers, President of the Nickel Institute
A chemist by training, Véronique Steukers is now the first woman to head the global organization of nickel producers, the Nickel Institute. Her career path has taken her far from the laboratory and into the heart of an industry facing significant environmental, industrial, and social challenges. We meet her.

What makes nickel so essential today?
Nickel is a surprisingly versatile metal, valued for its strength and durability. This explains its presence in many everyday applications. It has been used for over 100 years in the manufacture of stainless steel, which can be found everywhere in our kitchens, particularly in sinks and cutlery. Nickel makes these utensils highly durable and more resistant to corrosion, particularly from household products. It is also found in certain infrastructures. The Atomium, for example, was completely covered with a layer of nickel-containing stainless steel after its original coating had aged. This guarantees several decades of resistance without degradation. Nickel is also essential for electric car batteries because it improves their energy density, as well as for many other renewable energy technologies. Finally, it is one of the most recycled metals and its importance is set to grow with the increasing return of end-of-life batteries.
What is the role of the Nickel Institute?
The Nickel Institute is a global organization based in Canada with offices on several continents. Its main mission is to promote responsible nickel sourcing and support the sustainable development of this industry. To this end, we have three complementary departments. The first, scientific, is composed of toxicologists specializing in human and environmental health, as well as a prevention advisor responsible for worker protection. The second focuses on public policy and sustainability by monitoring developments in international regulations. It develops methodologies, particularly for measuring carbon footprints. Finally, the third is dedicated to market development and ensures that the various applications of nickel are better known and that markets remain open globally.
You have just been appointed president of the Nickel Institute. What does this appointment mean for your career as a chemist?
It's a role I really wanted to achieve in my career. After studying at UNamur, spending a year studying industrial chemistry in Leuven, completing a PhD in England and a post-doctorate in the Netherlands, I no longer practiced chemistry in a laboratory, but my training has always accompanied my career. It gave me an analytical and critical mind, an understanding of chemical substances, their properties, and industrial processes. This has enabled me to communicate effectively with authorities and stakeholders throughout my career. I often explain that studying chemistry does not only lead to a career in the laboratory. It opens many doors and gives access to a multitude of career paths.
What environmental and societal challenges await the nickel industry?
The stakes are high, especially because this metal is essential to the energy transition. It is found in many climate-related technologies, such as electric car batteries, hydroelectricity, and wind turbines. But nickel remains a mining product, and its production is mainly located in developing countries where the environment, working conditions, and local communities are not always a priority. That is why the Nickel Institute works closely with authorities, companies, and other stakeholders to improve understanding of nickel and its risks. The goal is to ensure that those who produce or handle this metal are aware of best practices for managing the risks associated with extraction, production, and industrial use. The challenge, therefore, remains to guide the industry toward more responsible and sustainable practices.
What do you remember most about your time at UNamur?
I loved my years at UNamur. I am Flemish, and many of my friends did not understand why I chose Namur over Leuven, but I have never regretted it. The atmosphere was very friendly, and the professors and teaching assistants were very welcoming. So much so that we are still in touch, forty years after our first year studying chemistry!
Is there a particular memory you would like to share?
One of my fondest memories is the series of concerts we organized at the Arsenal for two years in a row. I played the piano and had the opportunity to perform a piece for four hands with Professor Jean-Marie André, as well as a trio with two other professors. It was something that came very naturally in Namur, thanks to the small size of the university, where everyone knew each other. I'm not sure I would have found that same closeness anywhere else.
What advice would you give to future chemists?
Don't hesitate! Studying chemistry opens many doors. Of course, you need to have a scientific mind, but it's a course of study that allows you to develop skills that are useful in many professions. I would also advise placing real importance on languages. In Belgium, mastering several languages is an essential asset for advancing in industry. I also notice that there are many more opportunities for women in scientific careers than there used to be. At the beginning of my professional life, I was often the only woman in the room, but today teams are much more mixed, even in heavy industries such as metals. The fact that I became the first female president of the Nickel Institute is quite encouraging.

This article is taken from the "Alumni" section of Omalius magazine #39 (December 2025).

Did you know?
February 11 is International Day of Women and Girls in Science. To mark the occasion, UNamur is organizing the sixth edition of its Women in Science conference. This annual event aims to promote women's and girls' access to science and technology and their full and equal participation. It highlights the important role of women in the scientific community and is an excellent opportunity to encourage and promote equal opportunities for all genders in science and technology.



